Extraction and separation of chemical species
- Knowledge and skills
- Understand the principle of solvent extraction and hydrodistillation
- Learn how to use a data table to choose a solvent or develop an experimental protocol
- Learn about the risks and safety instructions to be followed when using organic solvents
I - Introduction:
When making tea, it is the taste and aroma contained within the leaves that are sought after. To obtain a good tea, it is necessary to transfer these substances from the leaves to the water (as they are soluble in water). A scientist would describe this process as the extraction of colorants and aroma from the tea using a solvent (water). Various techniques are available for this purpose. What are they? And how are they performed?
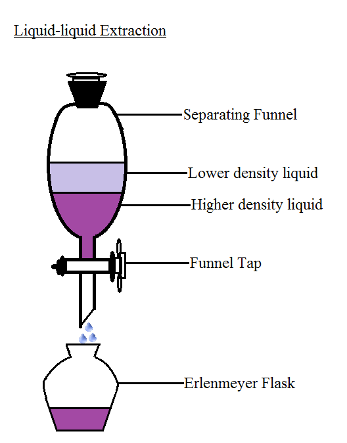
II - Liquid-liquid extraction:
Process
- Water (the initial solvent) and the chemical species to be extracted are placed in a separating funnel (aqueous phase).
- The extracting solvent is added (organic phase).
- The separating funnel is closed, agitated to mix the contents, and then opened to release any gas.
- The separating funnel is set aside, allowing the two phases to separate through decantation:
- The organic phase contains the extracting solvent and the extracted chemical species (The chemical species is more soluble in the extracting solvent than the initial one).
- The aqueous phase contains the initial solvent.The denser phase settles at the bottom of the separating funnel, while the less dense phase rises to the top
- Both solutions are carefully collected in separate beakers.
III- Solid-liquid extraction:
Step 1 - Direct solvent extraction
Direct solvent extraction involves dissolving a solid by allowing it to pass into a liquid, either through maceration, decoction, or infusion:
- Maceration: This operation entails soaking a substance in an open vessel filled with a liquid to extract soluble components. Example: pickles
- Infusion: It’s a method of extracting active compounds or aromas from a plant by dissolving them in an initially boiling liquid.
- Decoction: This method extracts active compounds and/or aromas from typically harder plant parts (e.g., roots, seeds, bark, wood) by dissolving them in boiling water.
Step 2 - Filtration

One can use a simple filter to separate solids from liquids.
Note : Regular filtration takes time. In laboratories, chemists and biologists tend to use a more complex yet fast type of filtration: the vacuum filtration (Büchner filtration)
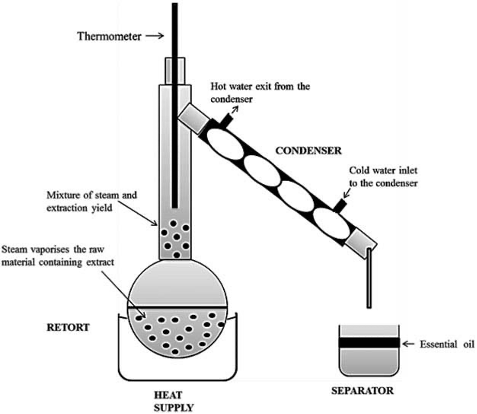
IV- Hydrodistillation:
Step 1 - Extraction of essential oil:
This involves the distillation of a mixture of water and a natural product placed in a flask.
When heating this mixture, the aromas from the natural product are carried away by the steam. To recover these aromas, the vapors are condensed using a cooler (refrigerant/condenser), allowing the recovery of the aromas. The distillate obtained in the test tube consists of two phases: an organic phase containing the essential oil and an aqueous phase containing water.
Step 2 - Liquid-liquid extraction:
A liquid-liquid extraction process is carried out to recover the essential oil using a separating funnel.
Fichier introuvable : Extraction and separation of chemical species.html